












The effect of salts in old buildings is a very important yet very overlooked topic in practical building conservation. Salts play a very important role in wetting-drying processes. They significantly affect the breathability of old buildings, acting as a chemical moisture barrier preventing evaporation.
In this section we are going to discuss in depth the effects of salts and how they affect old buildings. Understanding the effects of salts is very important because they profoundly affect the condition of the masonry. Old walls with high salinity must also be renovated differently from walls with low to moderate salinity.
Here is a video explaining and demonstrating some of these concepts.
Salts are a wide range of minerals consisting of two electro-chemical parts called ions. The electrically positive part is normally a metal, commonly Na+, Ca+, K2+ and Mg2+. The electrically negative part are other chemical groups, most commonly chlorides (Cl-), nitrates (NO3-) and sulphates (SO42-).
In various combinations, these 7 ions produce 4 x 3 = 12 elementary salts, which account for over 98% of the salts found in old walls.
Table salt (NaCl) is just one of the many types of salts.
In dry state, salts are organized in a 3D-lattice, the solid salt crystals being held together by intermolecular electrical attraction forces. Small crystals are white or yellowish powders which can have a salty or bitter taste.
In the presence of water, however things change. Salts become dissolved by water: the salt lattice gets surrounded by charged water ions (positive H+ and and negative OH- ions) pulling the salt lattice apart, breaking down its organized crystalline structure. The positive and negative salt ions become separated, travelling independently with the liquid water into the masonry, but retaining their individual positive or negative charges in the solution.
Once humidity evaporates, salts re-crystallize, forming the 12 common salts.
The most common types of salts found in the fabric of old buildings are:
Chlorides are plain sea salts (e.g. common table salt). Sea salts are transported by wind and fog inland ending up in the ground or washed into the buildings.
Salts sprinkled onto the roads during winter are chlorides that end up in the ground then absorbed into the building fabric.
Nitrate salts are predominantly originating from the decomposition of organic waste matter. Churches, chapels, graveyards and former battlegrounds contain a large amount of nitrates.
They are also prevalent in farming areas. Animal- and farming by-products such as manure or fertilizers contain high quantities of nitrates, which have been affecting that old farm building or barn walls for centuries. When these buildings are converted to dwellings the high salt content of the walls must be known and mitigated.
Sulphates are one of the most dangerous or damaging type of salts as they crystallize in long needle-shape crystals which can cause a lot of damage to the old building fabric.
Sulphate salts can be found in modern cement, added to it as an additive to make cement set slower. Under certain conditions the sulphates in cement can react with other salts in the environment, resulting in extreme salt expansion known as sulphate attack, causing severe damage to concrete structures and plasters.
Sulphates are also formed after combustion, being abundantly present in the chimney soot or in emission gases of the polluted atmosphere. When airborne sulphates are washed-down by rainwater, the resulting acid rain containing weak sulphuric acid, dissolves the lime, damaging old limestone statues or façades.
The presence of these salts can be measured and assessed with specialist tools. We do this on a regular basis as part of our professional building surveys.
In very small quantities, salts can natively be present in bricks. Bricks, made of clayey soil, naturally contain small amounts of salts. However, most salts are deposited over time into the building fabric from various sources.
Here are the most common sources of salts:
The effect of salts or the contribution of salts to the decay of the masonry is not obvious when looking at an old, decaying, damp wall. In such cases it is assumed that the cause of the problem must be high humidity and/or potential frost damages. Because an old wall is subject to all these factors - high humidity, frost, salinity - throughout the years, it is very difficult to quantify the individual effect of these factors.
However, studying the problem of wetting and drying under controlled lab conditions allows one to pull this multi-variable problem apart and understand the effect of each variable over the old masonry. Here are some of the experiments we have done and some of the important findings that we learned.
We have taken two identical "old-style" heritage bricks, one salty (submerged in sea water, then dried out thoroughly), the other one not salty. In the lab the two bricks have been subjected to ongoing wetting and drying cycles using a humidifier and a dehumidifier.
Both bricks have been subjected to high moisture content (90-95% RH) using a humidifier, then the moisture has been reduced to normal levels using a dehumidifier. Each wetting-drying cycle has lasted for 2 hours, and the cycles have been repeated non-stop, 12 times a day, 7 days a week, for several months. After about 3 months and 1,000 wetting-drying cycles we have observed some interesting changes in the bricks.
The non-salty brick, despite being subject to ongoing very high humidity, stayed perfectly intact.
The salty brick - subject to high humidity and salts - started spalling and crumbling badly, leading to significant fabric damage as shown in the photo gallery below.
These findings explain what we see in real life:













Salts have a profound affect onto old masonries, affecting them in many ways. Yet, their effects are commonly understated or mistakenly attributed to the presence of moisture.
Here are some of the most important effects of salts:
When humidity levels decrease or the temperature increases, salts crystallize. During crystallization salts expand in volume by 5 - 10 times (500% to 1,000%). The expansion generates enormous forces inside the masonry (up to 800 atmospheres), capable of breaking down even concrete. The high crystallization pressure results in the breakdown of paint, plaster and historic masonry, leading to crumbling, spalling, flaking or cracking of the building fabric.
Salt crystallization is similar to water freezing into ice. Ice crystals "only" expands in volume by about 10% yet the induced crystallization pressure can crack iron or steel pipes. In comparison, salts expand many times, thus capable of creating significant damages.
Salts are the primary reason behind the irreversible damage and loss of historic building fabric - not water. Salts are the main reason behind the damage and breakdown of plasters, why old buildings need to undergo a major replastering cycle every few years.
Here are some examples of salt damages, different buildings and walls being damaged by salts.































Every time when ambient humidity increases or the temperature decreases for whatever reason (e.g. wet season, period of rain, day-night variations etc.) salts tend to absorb moisture from the ambient air (hygroscopic action) and liquefy.
One salt molecule in a humid environment can attract and hold many-many water molecules. A well-known example of this is the behaviour of table salt in a salt shaker which in a high-humidity environment goes soft and sticky from the absorbed moisture from the surrounding air.
As a result salty bricks and plasters retain much more humidity than non-salty ones. Lab experiments have demonstrated that the moisture content of salty bricks (green) can easily be about 4.5 times higher than of non-salty bricks (blue), making salty masonries MUCH damper than non-salty ones, under the exact same conditions.
As most salts in an old wall, due to evaporation, get deposited on the surface (known as efflorescence) or just under the surface (subflorescence), in a high-humidity environment salts can make a wall fabric look damp even if the wall in depth is significantly drier.
This brings up an important technical point about the re-plastering of old, salty walls. The old, salt-laden plaster MUST be removed completely and affected wall sections must be fully re-plastered in order to ensure a salt-free = dry surface.
Retaining an old salt-laden plaster and just skimming it over in order to save money is a common and often costly renovation mistake which one can easily avoid by understanding what salts do and how they behave. A salt-resistant lime base coat must also be used to protect the longevity of the plastering.
Just as salts absorb humidity from the environment, they also won’t release it easily when ambient drops or when the wall starts drying out. In comparison to freshwater wetting, it takes a lot more energy to make water evaporate from a salty wall fabric.
In our lab experiment a non-salty brick needed 2 days to become dry, while a salty brick needed 8 days (4 times longer) to dry under the exact same circumstances. This can be seen on the above graph, where the "flattening" time of salty brick (green) is much longer than of the non-salty one (blue).
By hygroscopically trapping the evaporating moisture, salts act as an energy moisture barrier, having a similar behaviour to cement or other physical moisture barriers.
There is another important, less known aspect of wet salts. Salts are electrolytes (wet solutions with an electric charge) that make the wall fabric electrically conductive. A non-salty wall fabric is an insulator. A salty wall-fabric becomes a good conductor.
Measurements have shown that the electrical resistance of non-salty and salty walls differs significantly. While a non-salty wall fabric (red) has a resistance of hundreds of mega-ohms (being an insulator), the electrical resistance of salty walls (grey) can be 100 to 10,000 times less, decreasing steeply with the increase of salinity and humidity.
Why is this important? Because electrical resistance influences other chemical-physical properties of the wall fabric. Electromagnetic (EM) fields in any conductor can induce energy. For an old wall this translates to the generation and existence of small voltages and currents in the fabric. In other words, an electrically conductive wall fabric becomes susceptible to various EM phenomena from the environment.
The induced electrical charges interfere with the movement of water molecules and salt ions inside the capillaries, affecting wetting and drying cycles. Electrical phenomena in damp masonries are discussed in much more detail here.
Measuring the type and concentration of salts in the mortar or wall fabric is essential for identifying the source of moisture affecting the structure. Different salts originate from different sources—such as the ground or surrounding building materials—allowing us to determine which moisture source is most influential.
A relatively easy practical way to detect and quantify these salts is by using chemical indicator strips, which change colour when exposed to specific salts or materials. We focus on three key salts—sulphates, nitrates, and chlorides—as they account for over 95% of salt-related damages.








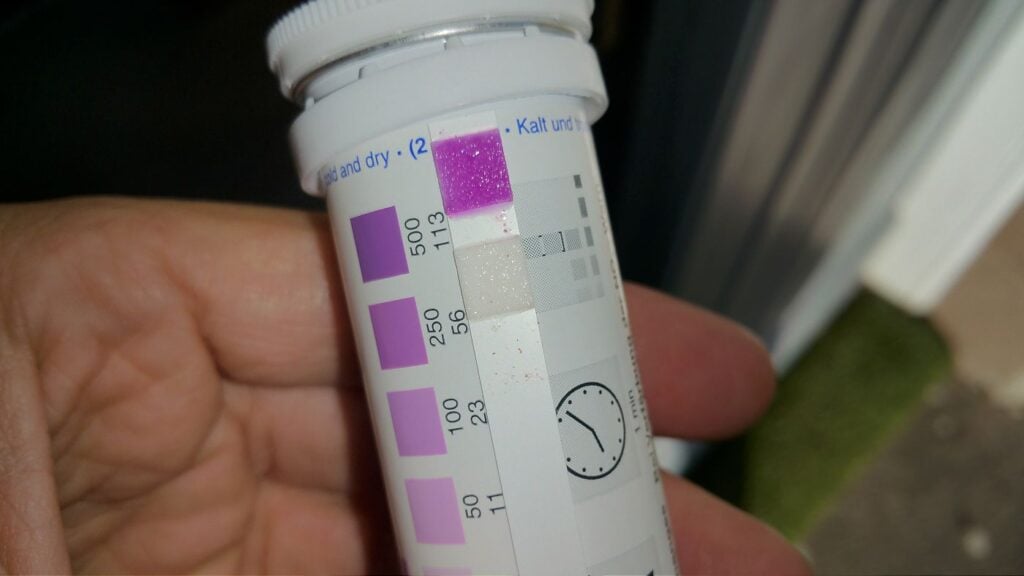

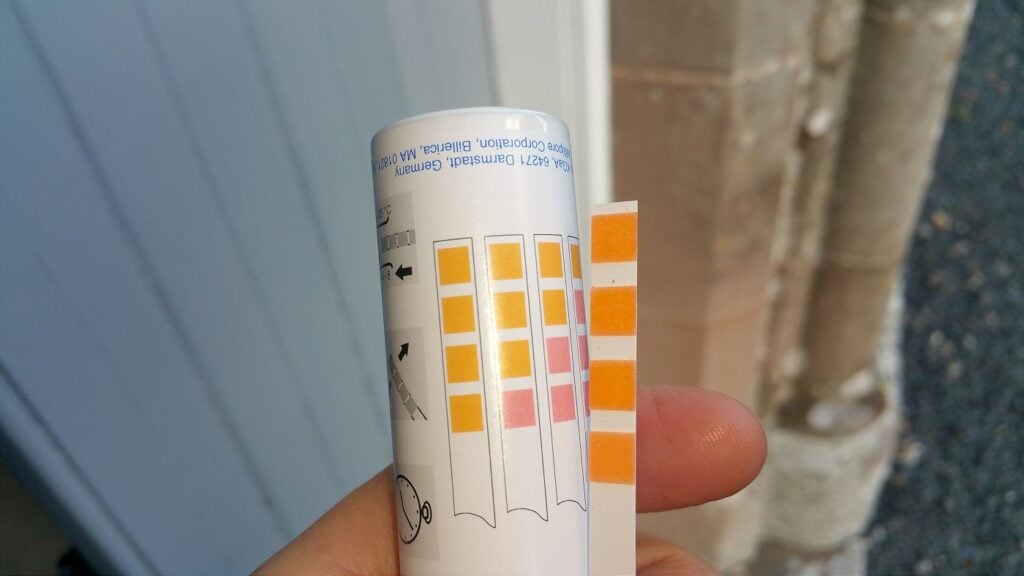



As the building ages, more and more salts accumulate inside the building fabric, leading to higher and higher salt concentrations. The salts have become part and parcel of the building fabric and can't be easily removed from it.
One of the most common mistakes during the renovation of old buildings - especially during farm or barn conversions - is not being aware of the potential problems salts can create. Some common problems found in old farms and barns are highlighted by the video below.
Various desalination procedures have been developed, but these tend to be complex. As a result most salt problems are managed by using the right type of plasters.
Replastering an old salty wall with a breathable general purpose lime plaster is not a long-term solution. In the presence of high salinity, the fresh lime plaster is usually short lived as it will be broken down by the crystallizing salts found in abundance in old farm buildings.
The right solution is using special salt-resistant lime plasters. The technology of these plasters originates from ancient Rome. The Romans figured out that if the soft lime is mixed with volcanic sands and ashes from Mount Vesuvius it will result in salt-resistant lime mixes that can even withstand sea water for many decades. These pozzolanic lime plasters have been used in Venice for centuries, performing extremely well in damp and harsh environment.
Applying a salt-resistant lime base coat under the main lime coat will extend the life expectancy of your plaster by about 10X, making your lime plaster last much-much longer, without sacrificing breathability in any way.
Feel free to reach out to us with any questions.
The destructive effect of salts onto old buildings is of great concern and as such it is being researched by many universities worldwide. For those interested in the research data, here are some international research papers about the importance of salts in building conservation, the salt crystallization process and their effects onto old masonry.
Here are some other related pages that you might want to read to broaden your knowledge in this field.
Here are some practical solutions related to this topic:
Here are the some recommended materials / products that can help solving or dealing with some of the problems discussed on this page.
Here are some of our projects where we have dealt with some of the issues discussed on this page:
Here are some photos demonstrating these concepts. Click on any image to open the photo gallery.
Most of the time the presence of salts is a more serious problem to old masonries than water. Understanding the type and concentration of salts in the masonry can help one to better understand the source and nature of moisture that is affecting the building. Here are some photos about the salt tests we perform on site.
Here is an 18 metre tall 200 year-old listed wind mill, close to the sea. Wind driven rain created major problems, resulting in ongoing water leaks. The building has been sympathetically waterproofed and thermally insulated with lime plasters only, using breathable heritage-friendly materials only.
This 150 year-old Victorian house has been replastered with modern building materials (cement and gypsum plasters). 3 months after the replastering the paint started to go. Another 3 months later the walls had to be fully replastered due to salt related damages.